Introduction
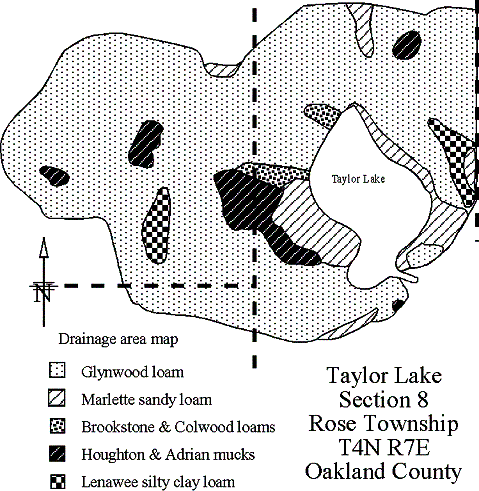
Watershed
Sampling
Oxygen
Chlorophyll a
Secchi
Phosphorus
Nitrogen
Alkalinity
pH
Conductivity
LWQI
Sediments
TAYLOR LAKE
ROSE TOWNSHIP
OAKLAND COUNTY
MICHIGAN
1984-2002 WATER QUALITY STUDIES
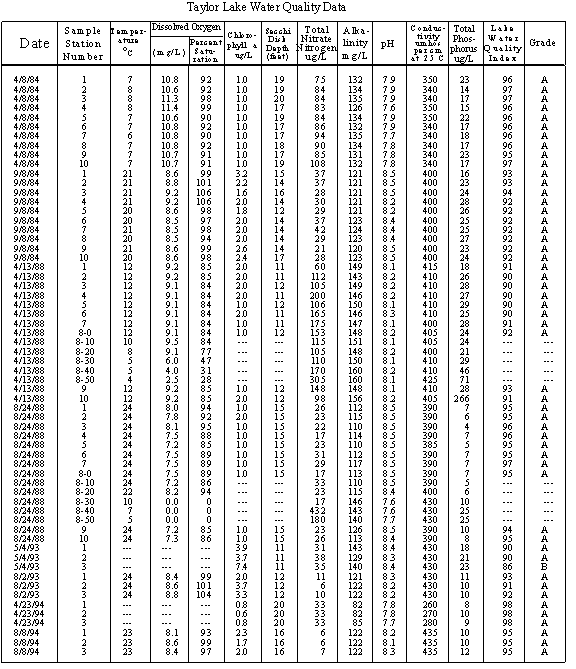
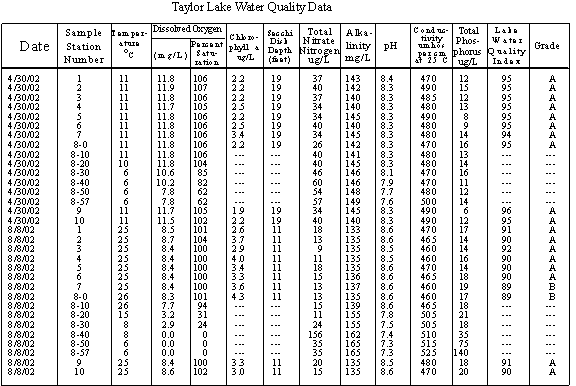
TAYLOR LAKE DATA
Taylor Lake is
a 35-acre natural moderately hard-water kettle lake located in Section 8,
Rose Township (T4N R7E) Oakland County, Michigan. The
lake has a maximum depth of 63 feet, a mean depth of 27.1 feet and contains
948-acre feet of water. The length of the shoreline
is 5491 feet. There are no islands in the lake.
There are no inlets.
The outlet is on the southeast corner. Water
from the Taylor Lake outlet joins the Shiawassee River south of Holly. The Shiawassee River joins the Tittabawassee River
south of Saginaw, forming the Saginaw River. The
Saginaw River flows into Lake Huron at Bay City.
The elevation of
the lake is 996 feet above sea level. The size
of the watershed, which includes all the lands that contribute water to
the lake, but does not include the lake, is 323 acres.
The drainage area, which includes the watershed and the lake,
is 358 acres. See map below.
The watershed to lake ratio is 9.23 to 1, which is on the
high side of normal for a Michigan inland lake. The
lake flushes about once every 2.8 years on an average
The latitude (42°
45.356N) and longitude (83° 39.697W) is the approximate location of
63-foot deep hole (Station 8).
THE WATER QUALITY STUDY
During certain
periods of the year, Michigan lakes have poorer water quality than the remainder
of the year. The water quality sampling is designed
to look at two of those poor water quality periods, one in early spring
when phosphorus which may released from the bottom sediments is distributed
throughout the water column by spring mixing, and a second in late summer
when the water is warmest, and the lake is stratified
(if it stratifies). During
most of the remainder of the year, the water quality is better. Thus if the lake gets high marks for water
quality during early spring and late summer, it probably has pretty good
water quality all year long.
SAMPLE DATES
Dr Chuck Cubbage and his Wayne County Community College class collected 10 surface samples for water quality testing in both spring and summer 1984. Top to bottom temperature and dissolved oxygen profile data were collected in summer. The lake was mapped by Dr. Cubbage and his students at this time
WQI limnologists collected ten surface samples plus top to bottom samples every 10 feet for water quality testing April 13, 1988, August 24, 1988, April 30, 2002 and August 8, 2002. Top to bottom temperature and dissolved oxygen profile data were collected each time the lake was sampled. Bottom sediment samples were collected at the ten in-lake stations in 1988 and again in 2002.
Tim Green collected three spring surface samples for water quality testing May 4, 1993 and April 23, 1994. WQI limnologists collected the same three surface samples for water quality testing August 2, 1993 and August 8, 1994
Secchi disk data have been collected on Taylor Lake since 1974. However the 1975-1979, 1981-1982, and 1994-1997 data are missing.
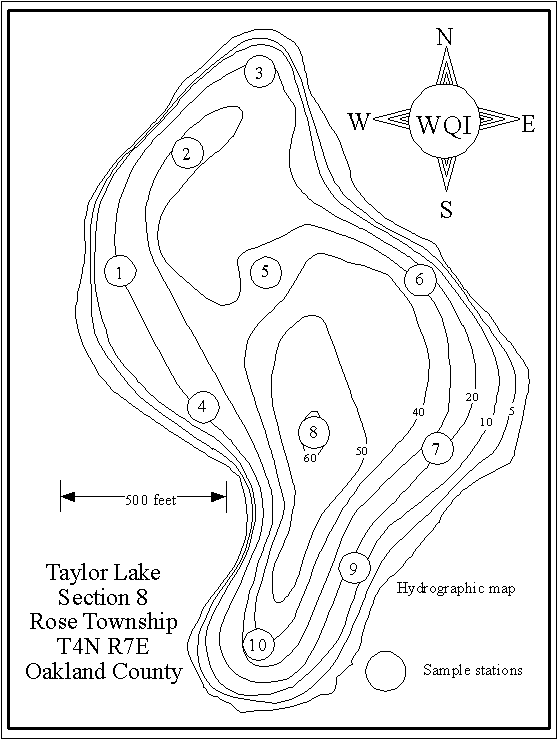
THE SAMPLE STATIONS
The locations of
the 1984, 1988 and 2002 sample stations are shown as circles on the map below. The three sites sampled in 1993 and 1994 were Station
1 on the north end, Station 2 over the deep hole and Station 3 on the south
end
THE ANALYSES
Dissolved oxygen,
temperature, and Secchi disk transparency measurements were conducted in
the field. Total phosphorus, conductivity, alkalinity,
total nitrate, chlorophyll a, and pH tests and bottom sediment analyses were
performed in the Water Quality Investigators laboratory near Dexter, Michigan. All tests followed the procedures outlined in Standard Methods for the Examination of Water and Wastewater,
1989.
THE DATA
Temperature and
dissolved oxygen data discussed below are found on the graphs. Other water quality data are found in the tables at
the end of this report and on the enclosed atlas pages.
TEMPERATURE AND DISSOLVED OXYGEN
Temperature exerts
a wide variety of influences on most lakes, such as the separation of layers
of water (stratification), solubility of gases, and biological activity.
Dissolved oxygen is the parameter most often selected by lake water quality scientists as being important. Besides providing oxygen for aquatic organisms to use, in natural lakes, oxygen is involved in phenomena such as phosphorus precipitation and release from the lake bottom sediments and decomposition of organic material in the lake.
1984
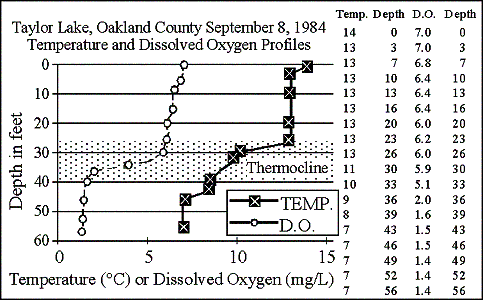
The temperature and dissolved oxygen profile graph shows in late summer
1984 the lake formed a 13-foot thick thermocline (defined as a layer of
water in a lake where the temperature changes more than one degree Centigrade
per meter of depth, and shown shaded on the graphs) from 26 to 39 feet. Although
the dissolved oxygen concentration started to drop near the top of the thermocline,
the lake did not run out of dissolved oxygen at any depth.
1988
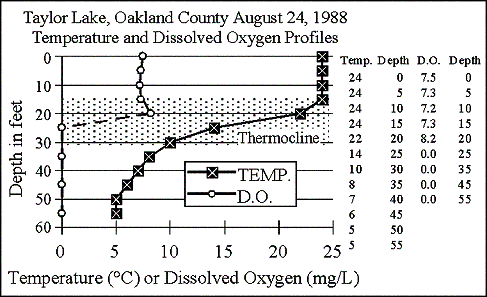
The graph shows
in late summer 1988 Taylor Lake formed a 15-foot thick thermocline from
15 to 30 feet. Above the thermocline, dissolved
oxygen was plentiful. Dissolved oxygen reached
a peak of 8.2 mg/L in the thermocline at 20 feet, probably the result of
an algal bloom that settled there. This year
the lake ran out of dissolved oxygen at 25 feet, and that condition remained
to the bottom. The hypsographic (depth-area)
graph shows about 52 percent of the lake is deeper than 25 feet.
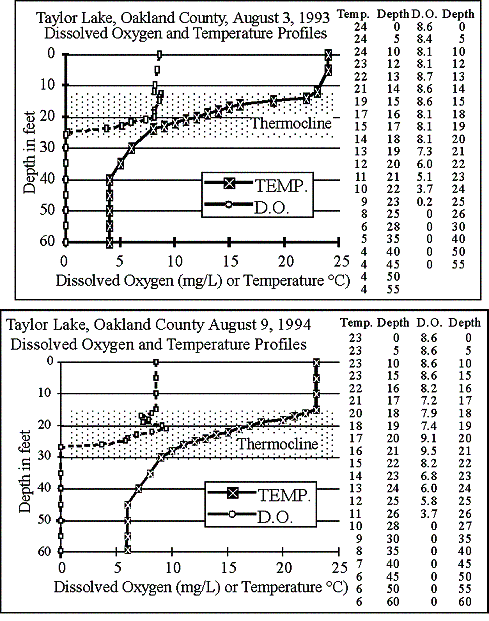
1993
The graph shows
in late summer 1993 Taylor Lake formed a 12-foot thick thermocline from
12 to 24 feet. Above the thermocline, dissolved
oxygen was plentiful. Dissolved oxygen dropped
to zero at 26 feet, and that condition remained to the bottom. About half the lake is deeper than 26 feet.
1994
In late summer
1994, a 15-foot-thick thermocline formed, from 15 to 30 feet. This year dissolved
oxygen again peaked in the thermocline, then reached zero at 27 feet, and
that condition remained to the bottom. About
50 percent of the lake is deeper than 27 feet.
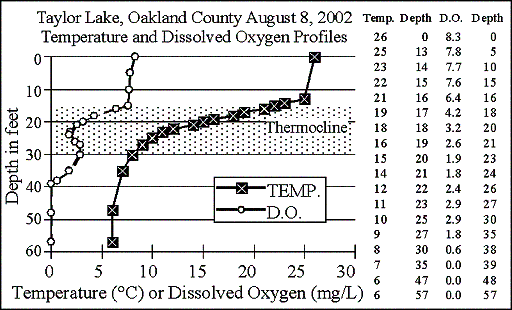
2002
The graph shows
in late summer 2002, Taylor Lake again formed a 15-foot-thick thermocline,
from 15 to 30 feet. Dissolved oxygen started
to drop at the top of the thermocline, to a low of 1.8 milligrams per liter
at 24 feet, then increased again to 2.9 milligrams per liter at 27 to 30
feet. It then dropped to zero at 39 feet. About 26 percent of the lake is deeper than 39 feet.
LATE SUMMER DISSOLVED OXYGEN SATURATION
Since dissolved
oxygen concentrations in water vary as the temperature varies, with cold
water holding more dissolved oxygen than warm water, dissolved oxygen saturation
is a better way to judge the amount of oxygen dissolved in the water. The graph of surface dissolved oxygen saturation
values shows most of the time Taylor Lake has dissolved oxygen concentrations
between 84 and 107 percent of saturation, which is good.
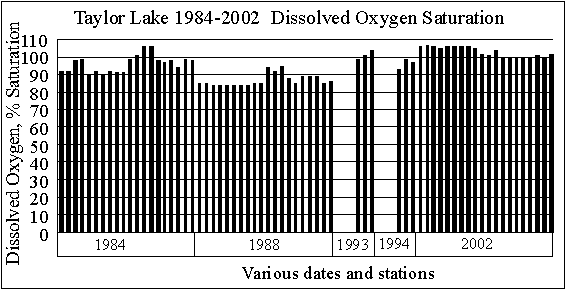
Low dissolved oxygen
concentrations (below 4 milligrams per liter) are generally insufficient
to support fish life. In most southern Michigan
lakes, there is no dissolved oxygen below the thermocline in late summer. (Since we study many Michigan lakes and measure the
oxygen depletion below the thermocline in many of them, we are not sure
why our depth sounder (and fish finder) shows the presence of fish in the
areas with no dissolved oxygen.)
However, as a limnologist,
I like to see some dissolved oxygen in the bottom water of a lake, even
if it is almost zero. This is because as long
as there is some dissolved oxygen in the water at the bottom of the lake,
phosphorus precipitated by iron to the bottom sediments will remain there. Once a lake runs out of dissolved oxygen in the water
at the bottom, iron comes back into solution, and when that happens, it
releases the phosphorus back into the water. This
can cause additional algae to grow when the lake mixes.
CHLOROPHYLL A
Chlorophyll a is
used by lake scientists as a measure of the biological productivity of
the water. Generally, the lower the chlorophyll
a, the better. High concentrations of chlorophyll
a are indicative of an algal bloom in the lake, an indication of poor lake
water quality.
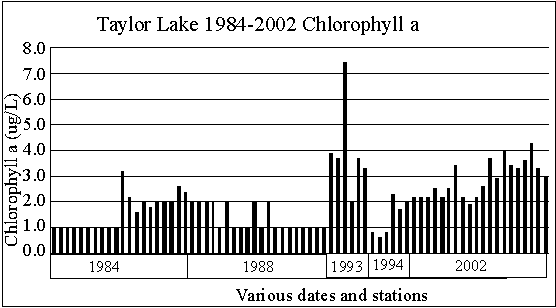
The highest surface
chlorophyll a concentration found by this writer in a Michigan lake was
216 micrograms per liter. Best is below one
microgram per liter.
Taylor Lake 1984
through 2002 chlorophyll a concentrations range from 0.6 to 7.4 micrograms
per liter. The individual high values are not
of concern.
However, the graph
seems to show chlorophyll a concentrations are increasing. Let’s hope that trend doesn’t continue.
SECCHI DISK TRANSPARENCY (originally
Secchi’s disk)
In 1865, Angelo
Secchi of Rome, Italy devised a 20 centimeter (8 inch) white disk for studying
the transparency of the water in the Mediterranean Sea.
Later an American limnologist (lake scientist) named Whipple
divided the disk into black and white quadrants which many are familiar
with today.
The Secchi disk transparency is a lake test widely used and accepted by limnologists. The experts generally felt the greater the Secchi disk depth, the better quality the water. However, one Canadian scientist pointed out acid lakes have very deep Secchi disk readings. (Would you consider a very clear lake a good quality lake, even if it had no fish in it? It would be almost like a swimming pool.) Most lakes in southeast Michigan have Secchi disk transparencies of less than ten feet. On the other hand, Elizabeth Lake in Oakland County had 34 foot Secchi disk readings in summer 1996, evidently caused by a zebra mussel invasion a couple of years earlier.
Most limnology
texts recommend the following: to take a Secchi
disk transparency reading, lower the disk into the water on the shaded side
of an anchored boat to a point where it disappears.
Then raise it to a point where it’s visible.
The average of these two readings is the Secchi disk transparency
depth.
We do it slightly
differently. We lower the disk on the shaded
side of an anchored boat until the disk disappears, and note the depth,
then report the depth to the next deepest foot. For
example if the disk disappears at six and a half feet, we report the Secchi
disk depth as 7 feet. The reason we do this
is that some suggest using a water telescope (a device that works like an
underwater mask and eliminates water roughness) to view the disk as it
disappears. Since we don’t use this device, we
compensate for it by noting the slightly deeper depth.
We feel it is only
necessary to report Secchi disk measurements to the closest foot. Secchi disk measurements should be taken between
10 AM and 4 PM. Rough water will give slightly
shallower readings than smooth water. Sunny days
will give slightly deeper readings than cloudy days.
However, roughness influences the visibility of the disk more
than sunny or cloudy days. Furthermore, it’s
been reported that most adults can see the Secchi disk disappear at about
the same depth, but grand-children see it disappear 3-4 feet deeper than
grand-parents.
If there are sample
sites where the lake is too shallow and the disk is visible when resting
on the bottom, the reading should be taken at a nearby deeper site. Since the sampling procedure is designed to obtain
"representative samples" moving the boat to an area where a Secchi disk
transparency reading can be properly taken is appropriate. In the case of Secchi disk readings, this procedure
is more valid than reporting the disk is visible on the lake bottom.
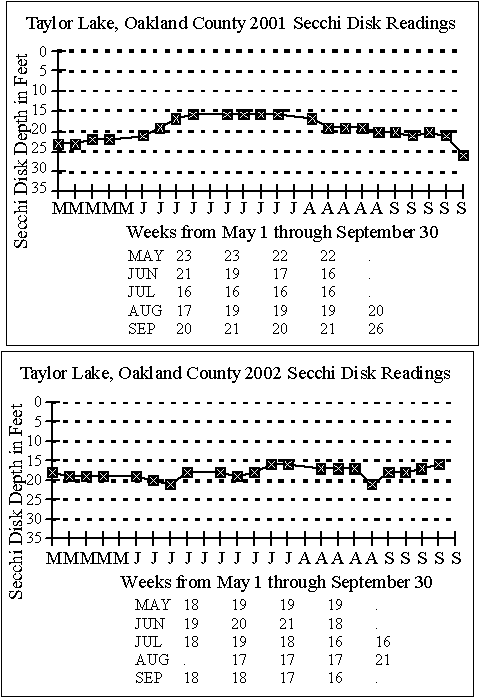
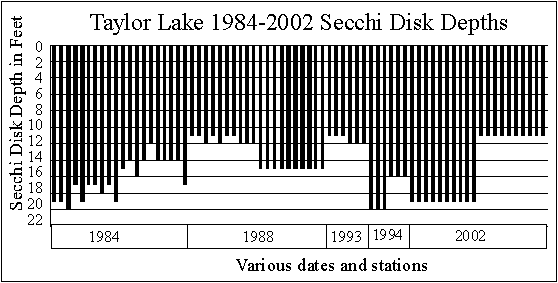
SECCHI DISK DATA
As noted earlier,
although quite a bit of Secchi disk data for Taylor Lake exists, there
are some gaps. Chuck Pilar did a good job taking
Secchi disk readings in 2001 and 2002.
The graphs below
show the Secchi disk readings collected by Pilar in 2001 and 2002.
In 2001, spring
data range from 21 to 23 feet. As the lake
warms in summer, the clarity drops to 16 to 17 feet (probably an algal
bloom), then the lake gets clearer (26 feet in October) in fall as the water
cools.
In 2002 Secchi
disk readings were much more uniform, ranging from 16 to 21 feet through
the warm months. This year the data graphed essentially
as a straight line, indicating the clarity of Taylor Lake didn’t change
as the water warmed from spring to summer. This
is a plus.
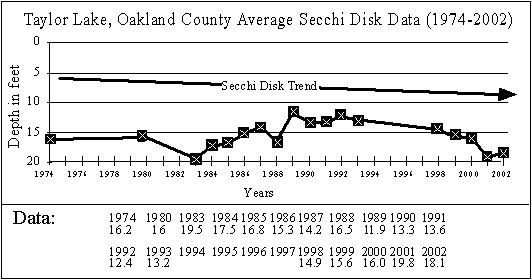
SECCHI DISK TREND GRAPH
Since Taylor Lake
residents have been taking Secchi disk readings on somewhat of a regular
basis since 1974 we were able to construct a Secchi Disk Trend Graph based
on averaged Secchi disk data for each year we have data for.
The Secchi Disk
Trend graph shows the average Secchi disk readings are generally between
13 and 18 feet, which is good. It also shows
the lake may be getting clearer, which is also good.
SECCHI DISK READINGS TAKEN WITH SAMPLES
The graph below shows the Secchi disk readings
collected when the samples were taken. The graph
does not show any specific trend, except that spring readings were deeper
than summer readings in 1984, 1994 and 2002. The
opposite was true in 1988 and 1993. The amount
of variation in the 1984 data is unusual.
Although there
are several forms of phosphorus found in lakes, the experts selected total
phosphorus as being most important. This is probably
because all forms of phosphorus can be converted to the other forms. Currently, most lake scientists feel phosphorus,
which is measured in parts per billion (1 part per billion is one second
in 31 years) or micrograms per liter (ug/L), is the one nutrient which might
be controlled. If its addition to lake water
could be limited, the lake might not become covered with the algal communities
so often found in eutrophic lakes.
However, based
on our studies of many Michigan inland lakes, we’ve found many lakes were
phosphorus limited in spring (so don’t add phosphorus) and nitrate limited
in summer (so don’t add nitrogen).
10 parts per billion
is considered a low concentration of phosphorus in a lake and 50 parts
per billion is considered a high value in a lake by many limnologists.
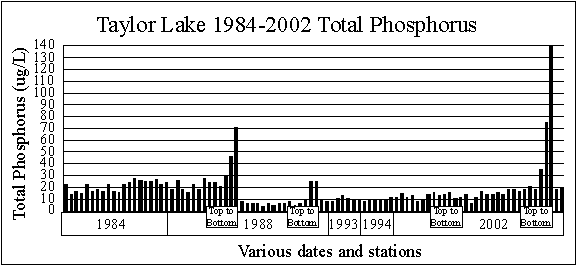
The graph of Taylor Lake total phosphorus concentrations shows the surface
phosphorus concentrations range from 4 to 29 micrograms per liter, but are
generally in the 5 to 15 microgram per liter range.
These are good phosphorus concentrations.
Three of the four top to bottom sample series show higher phosphorus
concentrations at the bottom of the lake. This
phosphorus is being released from the bottom sediments during anoxic (no
dissolved oxygen) conditions in the bottom water. The
graph also seems to show phosphorus concentrations may be increasing.
NITRATE NITROGEN
Nitrate, also measured
in the parts per billion range, has traditionally been considered by lake
scientists to also be a limiting nutrient. The
experts felt any concentration below 200 parts per billion was excellent
in terms of lake water quality. The highest
value found by this author was 48,000 parts per billion in a river which
flowed into an Ottawa County lake in Holland, Michigan
On the other hand,
we’ve studied hundreds of Michigan inland lakes, and many times we find
them nitrate limited (very low nitrate nitrogen concentrations), especially
in summer.
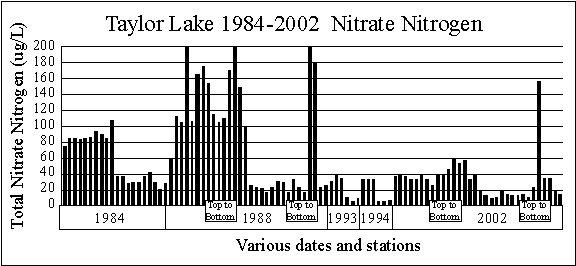
The graph comparing
the spring and summer nitrate-nitrogen concentrations from 1984 through
2002 shows spring surface values range from 33 to 200 micrograms per liter,
while summer values are much lower, ranging from 6 to 42 micrograms per
liter. These are normal nitrate nitrogen concentrations
for a Michigan inland lake.
The 1988 and 2002
top to bottom series of samples show higher concentrations of nitrate nitrogen
below the surface. We usually find this only
in high quality lakes.
We’re finding many
lakes have lower surface nitrate nitrogen concentrations in summer than
in spring. This is probably due to two factors. First, plants and algae growing in lakes as water
warms can remove nitrates from the water column. And
second, bacterial denitrification (where nitrates are converted to nitrogen
gas by bacteria) also occurs at a much faster rate in summer when the water
is warmer.
Generally limnologists
feel optimal nitrate nitrogen concentrations (which encourage maximum plant
and algal growth) are about 10-20 times higher than phosphorus concentrations. The reason more nitrogen than phosphorus is needed
is because nitrogen is one of the chemicals used in the production of plant
proteins, while phosphorus is used in the transfer of energy, but is not
used to create plant material. If the nitrate
concentration is less than 10-20 times the phosphorus concentration, the
lake is considered nitrogen limited. If the nitrate
concentration is higher than 10-20 times the phosphorus concentration, the
lake is considered phosphorus limited.
TOTAL ALKALINITY
Alkalinity is a
measure of the ability of the water to absorb acids (or bases) without
changing the hydrogen ion concentration (pH). It
is, in effect, a chemical sponge. In most Michigan
lakes, alkalinity is due to the presence of carbonates and bicarbonates
which were introduced into the lake from ground water or streams which flow
into the lake. In lower Michigan, acidification
of most lakes should not be a problem because of the high alkalinity concentrations.
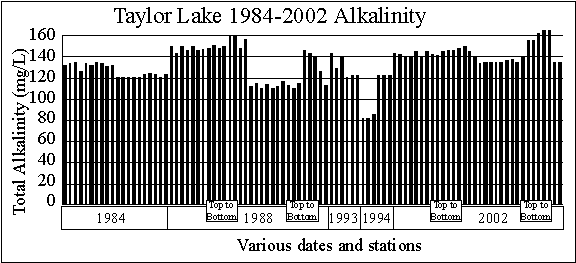
The graph of alkalinity
data shows three things.
First, the Taylor
Lake surface alkalinity data (82 to 156 milligrams per liter) indicate
Taylor Lake is a moderately hard-water lake, which is good. This is because hard water lakes have the
ability to precipitate some of the phosphorus that enters the lake to the
bottom sediments as calcium phosphate. This
pretty much ties up the phosphorus in the sediments.
Soft water lakes lack this ability.
Second, spring
alkalinities are higher than summer alkalinities, which is what we normally
see.
Third, alkalinites
appear to be increasing. That’s not a problem.
HYDROGEN
ION CONCENTRATION (pH)
pH has traditionally
been a measure of water quality. Today it is
an excellent indicator of the effects of acid rain on lakes. About 99% of the rain events in southeastern
Michigan are below a pH of 5.6 and are thus considered acid. However, there seems to be no lakes in southern
Michigan which are being affected by acid rain. Most
lakes have pH values between 7.5 and 9.0.
Taylor Lake pH
values (7.6 to 8.6) are within normal ranges for a high quality Michigan
moderately hard water inland lake. pH values
of the top to bottom samples shows pH generally decreases with depth. That’s normal.
SPECIFIC CONDUCTIVITY
Conductivity, measured
with a meter, detects the capacity of a water to conduct an electric current. More importantly however, it measures the amount
of materials dissolved in the water, since only dissolved materials will
permit an electric current to flow. Theoretically,
pure water will not conduct an electric current. It
is the perception of the experts that poor quality water has more dissolved
materials than does good quality water. We
concur.
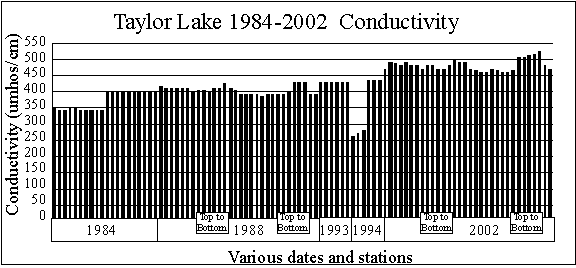
The graph shows
Taylor Lake 1984-2002 surface conductivity data (260 to 490 micromhos per
centimeter), which are normal for a moderately hard water Michigan inland
lake. The graph also shows conductivity in Taylor Lake may be increasing. Usually the source of salts in lakes is winter road
salting activities or salts from regenerating water softeners. The top
to bottom samples show in some cases conductivity (and therefore salt concentrations)
increase near the bottom. That’s normal.
THE LAKE WATER QUALITY INDEX
The Lake Water
Quality Index (LWQI) (Fusilier, 1982) used in this study to define the water
quality of Taylor Lake was developed for two reasons.
First, there was no agreement among lake scientists regarding
which tests should be used to define the water quality of a lake; and second,
there was no agreement among lake scientists regarding the meaning of the
data collected during lake studies.
Development of
the index involved two questionnaires which were sent to a panel of 555 scientists
who were members of the American Society of Limnology and Oceanography. The panel was specifically selected because they
were chemists and biologists with advanced degrees who studied lake water
quality.
The first questionnaire
asked the scientists to select tests which they felt should be used to
define lake water quality.
The tests most
often selected by the scientists became the index parameters (or tests). They were:
Dissolved oxygen (Percent saturation)
Total phosphorus
Total alkalinity
pH
Chlorophyll a
Temperature
Secchi disk depth
Conductivity
Total nitrate nitrogen
The second questionnaire,
sent out after the first was returned, asked the scientists what the results
of the tests they selected as good indicators of lake water quality meant.
After the responses
to the second questionnaire were tabulated, the nine tests and the accompanying
rating curves were combined into a Lake Water Quality Index.
The index ranges
from 1 to 100, with 100 indicating excellent lake water quality. The index rated lakes about the same way teachers
rate students: 90-100=A, 80-90=B, 70-80=C,
60-70=D, and below 60=E.
The highest index
for a Michigan lake studied by the author was Long Lake in Oakland County
at 100 in the spring of 1994. The lowest was
16 in an Ottawa County lake.
THE LAKE WATER QUALITY INDICES FOR TAYLOR LAKE
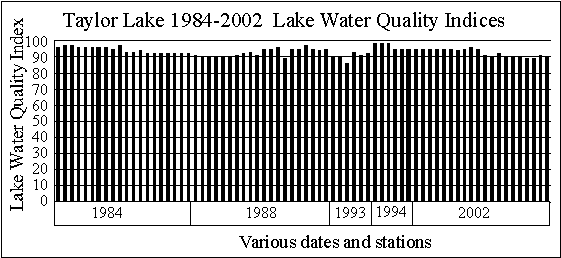
The graph shows
the spring and summer Lake Water Quality Indices for Taylor Lake from 1984
through 2002 are, with the exception of a spring 1993 sample and two summer
2002 samples, all 90 or above. These data indicate
the water quality of Taylor Lake was in the A range every time it was sampled. The reason the spring 1993 sample was below 90 (actually
86) was a high chlorophyll a concentration. The
reasons the two summer 2002 samples were less than 90 was chlorophyll a
concentrations, Secchi disk depths and pH values.
THE LAKE WATER QUALITY INDEX (LWQI) CALCULATION SHEETS
The Lake Water
Quality Index calculation sheets were developed to show graphically what
the results of the nine different lake water quality tests meant in terms
of lake water quality.
HOW TO READ THE LAKE WATER QUALITY INDEX CALCULATION SHEETS
Listed across the
top of the calculation sheets are the tests selected by the panel of experts
as being good indicators of lake water quality.
The figures which
look like thermometers are graphs which convert the test results (the values
found on the outside of the thermometer) to a uniform 0-100 lake water
quality rating (found on the inside of the thermometer).
The calculation
sheet combines all nine of the individual quality ratings into a single
Lake Water Quality Index. The index ranges from
1 (very poor lake water quality) to 100 (excellent lake water quality).
The index is portrayed
in three different ways: as a number ranging
between 1 and 100 in the circle marked LWQI, and by a color and position
on the sheet edge scale. The purpose of the sheet-edge
scale is to review quickly large numbers of lakes or sample sites within
a lake and determine how the quality of the various lakes or sites compare.
The position of
the red lines on the thermometer rating scales permits determination of
the parameter (or parameters) which cause the index to be depressed. The lower the red line, the greater the problem. A glance at the top of the problem rating scale identifies
the test and the test results. The rating scales
also permit determination of what test results would be considered excellent
in terms of lake water quality by the panel of experts surveyed. They are the numbers on the outside the thermometers,
near the top.
Since there was
not a big difference in the surface water quality at the ten stations in
either the spring or summer 2002, only two Lake Water Quality Index calculation
sheets are included in this report, one for the ten spring 2002 samples,
using averaged data, and one for the ten summer 2002 samples, using averaged
data.
The report marked
MASTER has all twenty 2002 Lake Water Quality Index calculation sheets. That is the only difference in that report and the
others. Lake Water Quality Index calculation sheets for earlier years were
included with those reports.
As an added note,
we’ve changed some of the rating curves in the Lake Water Quality Index
to more accurately portray high quality lakes. All
of the earlier LWQIs have been updated and recalculated for this report.
BOTTOM SEDIMENTS
Many times bottom
sediments tell us more about what is happening in a lake than the water
quality tests do. That’s because bottom sediments
provide sort of a history of what’s been happening in a lake, while water
testing just provides a snapshot.
Bottom sediments are collected with a Pederson dredge,
transferred to pint freezer containers and allowed to air dry. Once they are dry, the (usually) shrunken block of
material is measured to determine volume, then ground, placed in porcelain
dishes, dried at 100 degrees C, weighed, burned at 550 degrees C, and weighed
again. Color after air-drying and after burning
is also noted.
Bottom sediments
almost always come up from the lake bottom black, and many people consider
these black sediments “muck”. However that’s
not usually the case.
The bottom sediments
are black because no oxygen penetrates them, so the decomposition processes
which occur use sulfur rather than oxygen, and in this process, they produce
iron sulfides, which are black. However once
the sediments are exposed to air, they usually turn some other color.
If the sediments
remain black after air drying it usually means they are less than about
65 percent mineral (or more than 35% organic material).
Sediments also remain black if they are from soft water lakes,
but there’s a reason for that.
If the sediments
turn gray after air drying it usually means they are made up primarily
of carbonates. This is what we usually see
in moderately hard water and hard water lakes.
If the sediments
turn tan, it usually means they are made up primarily of clays. Further evidence of this occurs when we burn the
sediments at 550 degrees C.
We determine how
much bottom sediments shrink when they air dry because this information
is useful when considering dredging a lake. Normal shrinkage after air-drying
is in the range of 50 to 80 percent. However
sands and gravels don’t shrink at all. Excessive
shrinkage is more than 95 percent. In other
words, there is only five percent or less of the material remaining after
air-drying.
If the gray bottom
sediments remain gray after burning they are considered carbonates, and
the loss of material during this process is considered organic material. The results are expressed in the percentage of minerals
in the bottom sediments.
If the tan bottom
sediments turn red after burning, it means the lake is filling with clay. Clay enters the lake from near-lake activities such
as road building, home building or farming. Usually
clay is not a material that makes up the bottom sediments of most inland
lakes.
Highly organic sediments that remained black after
air drying usually turn tan after burning, but the mineral content is usually
quite low.
I consider high
quality bottom sediments from natural lakes to be above 85 percent mineral. And I consider bottom sediments less than 50 percent
mineral to be muck.
THE BOTTOM SEDIMENTS OF TAYLOR LAKE
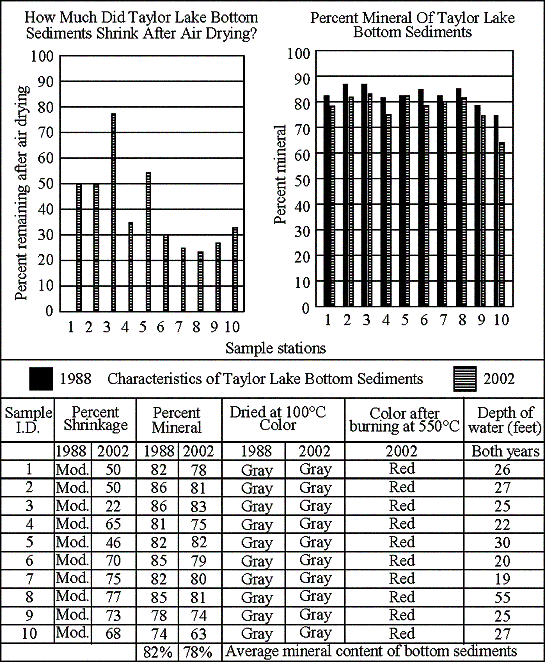
Bottom sediment
samples were taken at the ten in-lake stations in 1988 and again in 2002. Amount of shrinkage was not calculated in the 1998
samples.
None of the 2002
samples shrunk excessively (more than 95 percent is excessive). The graph shows the 1988 and 2002 data.
All 1988 and 2002
samples turned gray after air drying. That’s
good.
Color after burning
was not noted in 1988. In 2002 all samples
turned red after burning at 550 degrees C. The
red color indicates the presence of clay in the sediments. As noted above, clay is not a normal constituent
of Michigan inland lake bottom sediments. The
source of clays in lake bottom sediments is usually home building or road
building, or farming activities in the near lake areas.
The mineral content
of the 1988 samples ranged from 74 to 86 percent and averaged 82 percent.
The mineral content
of the 2002 samples ranged from 63 to 83 percent and averaged 78 percent.
These data indicate
organic material is building up at a faster rate than the lake can assimilate. Residents need to be aware of this and quit using
all fertilizers on near lake areas.
Wallace E. Fusilier,
Ph.D.
Consulting Limnologist
Dexter, Michigan
December 2002